

Arthrosamid
Book a Detailed Treatment Call
How does Arthrosamid work?
Arthrosamid is an intra-articular polyacrylamide hydrogel injection (iPAAG) which once injected into the knee’s inner joint cavity, restores viscosity within the synovial fluid. This improves the lubrication and provides cushioning in the joint that had naturally started deteriorated as a result of the osteoarthritis. The lubrication and cushioning provided by the injection significantly reduce pain for patients, all in one single dose solution. You can learn more about Arthrosamid injections from our partner website AMSK.
Why choose Arthrosamid treatment from London Cartilage Clinic?
Our exclusive service and injection protocol has been developed with the expertise and experience of Professor Paul Lee, who has carried out Arthrosamid injections on hundreds of patients. An industry leader in orthopaedic surgery, Professor Lee is a highly regarded joint and cartilage specialist with a proven track record of achieving life-changing outcomes for patients.
If you’re suffering with knee osteoarthritis and need an alternative option from conventional therapies to ease your symptoms and help you return to your daily activities without discomfort, an Arthrosamid injection from London Cartilage Clinic could be the ideal solution for you.




Frequently Asked Questions
Arthrosamid is an innovative and premium therapy for knee osteoarthritis, you can learn more about our pricing for this treatment on our price list
Arthrosamid differs from other viscosupplement injections like Hyaluronic Acid (HA), Platelet-Rich Plasma (PRP) and regenerative stem cell therapy. This is because Arthrosamid is a non-degradable hydrogel that has the ability to lubricate and cushion the joint for longer periods of time, which means longer-lasting pain relief for the patient.
Additionally, Arthrosamid can integrate into the synovial tissue in the joint capsule and clinical trials have shown that there were no serious side effects related to hydrogel. It is a safe and effective treatment for knee osteoarthritis with minimal side effects.
Arthrosamid has been a proven treatment for knee osteoarthritis for over 20 years, offering long-lasting pain relief to patients. Clinical trials of the injection were overwhelmingly positive, with over 70% of patients in one trial reporting that they had noticed improvements in their symptoms.
Studies have shown that the beneficial effects of Arthrosamid can last for up to a year. However, some research has suggested the effects can be even longer, potentially lasting several years. Trials are still ongoing to monitor the full extent and lasting impact of Arthrosamid on some patients for five years.
Osteoarthritis is a condition that can affect bones in many areas of the body and in turn, many types of soft tissue within key joints. Pain is typically a result of the condition reducing the cushioning around these joints. In the most extreme cases, arthritis causes joint cartilage to degrade leading to bones rubbing together. This often results in pain triggered by movement.
Very. The intra-articular polyacrylamide gel has been confirmed as safe for the intended use of the treatment – relieving the symptoms of arthritic pain. Adverse side effects may consist of swelling and slight pain in the weeks following the procedure.
 Cartilage
Cartilage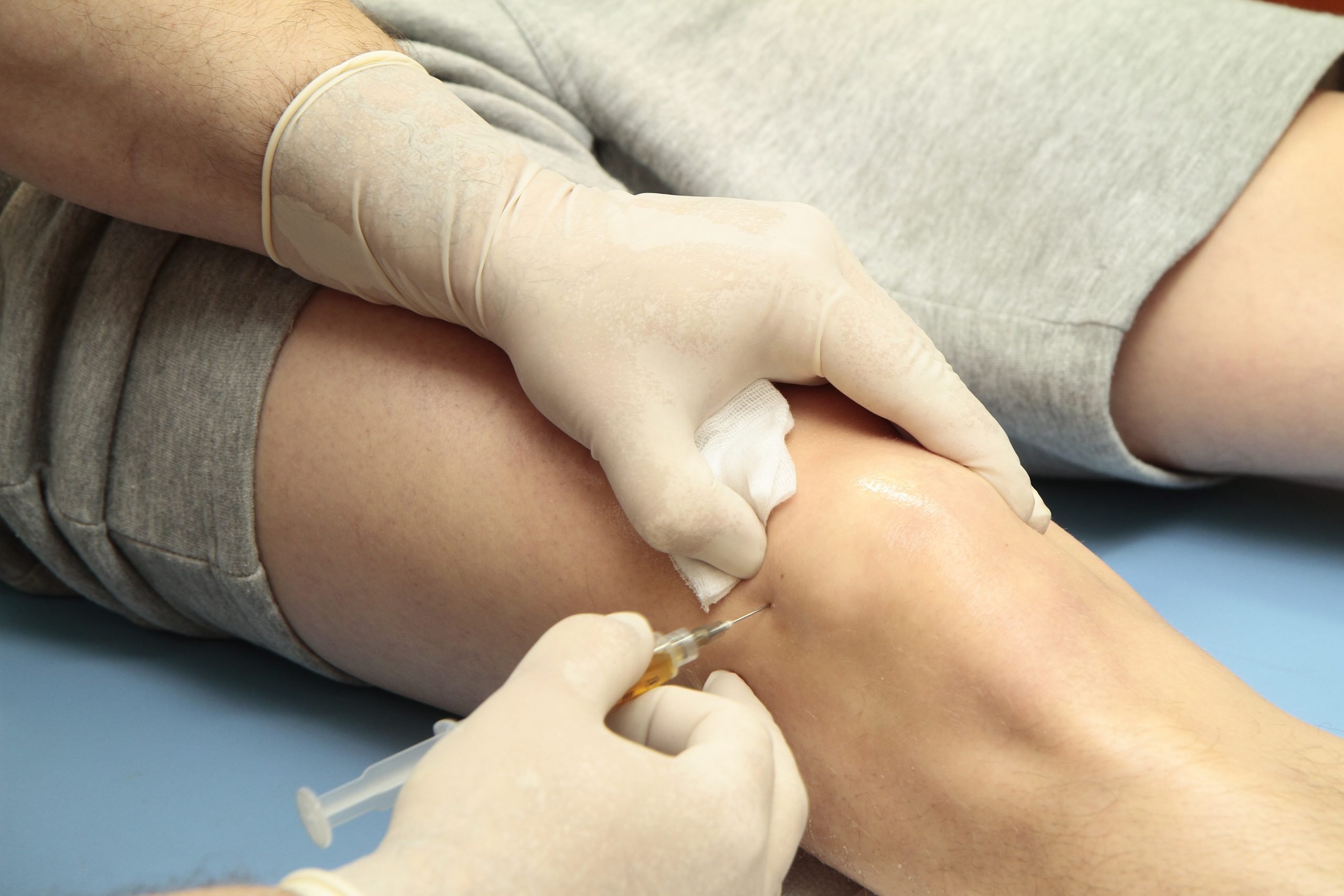 Knees
Knees
